
In general ligand apicophilicity increases with electronegativity and also with pi-electron withdrawing ability, as in the sequence Cl < F < CN.  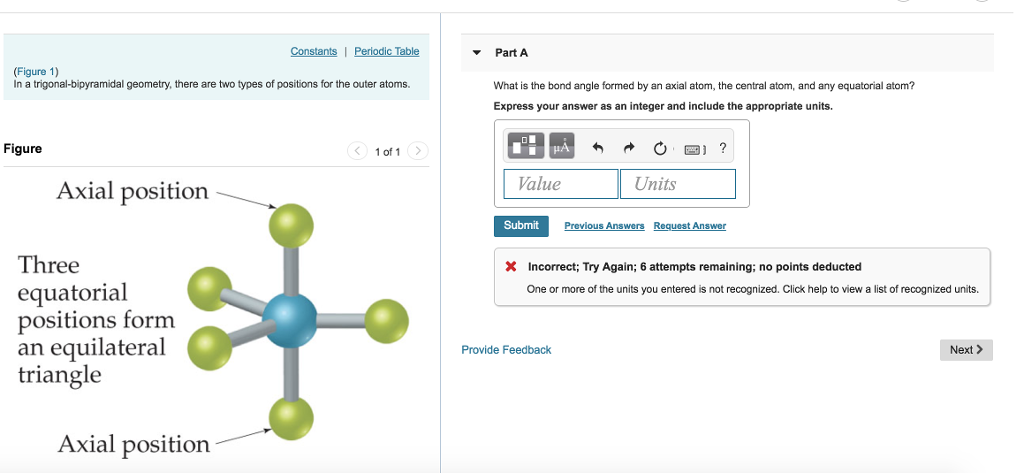
In the mixed halide PF 3Cl 2 the chlorines occupy two of the equatorial positions, indicating that fluorine has a greater apicophilicity or tendency to occupy an axial position. As examples, in PF 5 the axial P−F bond length is 158 pm and the equatorial is 152 pm, and in PCl 5 the axial and equatorial are 214 and 202 pm respectively. For molecules with five identical ligands, the axial bond lengths tend to be longer because the ligand atom cannot approach the central atom as closely. For phosphorus pentachloride as an example, the phosphorus atom shares a plane with three chlorine atoms at 120° angles to each other in equatorial positions, and two more chlorine atoms above and below the plane ( axial or apical positions).Īccording to the VSEPR theory of molecular geometry, an axial position is more crowded because an axial atom has three neighboring equatorial atoms (on the same central atom) at a 90° bond angle, whereas an equatorial atom has only two neighboring axial atoms at a 90° bond angle. The five atoms bonded to the central atom are not all equivalent, and two different types of position are defined. Axial (or apical) and equatorial positions Trigonal bipyramidal molecular shapeĮq = equatorial ligand (in plane perpendicular to unique axis) Examples of this molecular geometry are phosphorus pentafluoride ( PF 5), and phosphorus pentachloride ( PCl 5) in the gas phase. This is one geometry for which the bond angles surrounding the central atom are not identical (see also pentagonal bipyramid), because there is no geometrical arrangement with five terminal atoms in equivalent positions. In chemistry, a trigonal bipyramid formation is a molecular geometry with one atom at the center and 5 more atoms at the corners of a triangular bipyramid. Molecular structure with atoms at the center and vertices of a triangular bipyramid Trigonal bipyramidal molecular geometry
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
